Publication
Synergistic interplay of morphology and metabolic activity rule response to CAR-T cells in B-cell lymphomas
Y Chen, S Sabir, C Kuttler, J Belmonte, A Martinez, L Martin, L Lopez-Corral, A Martin-Sancho, JC Cañadas, C Montes, M. PTamayo, A Cedillo, P Balsalobre, P Barba, A Pérez-Martínez, VM Pérez-García
arXiv:2505.11894
MOLAB authors
Abstract
Cellular immunotherapies are one of the mainstream cancer treatments unveiling the power of the patient's immune system to fight tumors. CAR T-cell therapy, based on genetically engineered T cells, has demonstrated significant potential in treating hematological malignancies, including B-cell lymphomas. This treatment has complex longitudinal dynamics due to the interplay of
different T-cell phenotypes (e.g. effector and memory), the expansion of the drug and the cytotoxic effect on both normal and cancerous B-cells, the exhaustion of the immune cells, the tumor immunosupressive environments, and more. Thus, the outcome of the therapy is not yet well understood leading to a
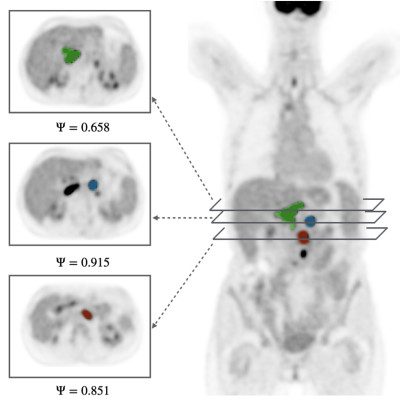
variety of responses ranging from sustained complete responses, different types of partial responses, or no response at all. We developed a mechanistic model for the interaction between CAR T- and cancerous B-cells, accounting for the role of the tumor morphology and metabolic status. The simulations showed that lesions with irregular shapes and high proliferation could contribute to long term progression by potentially increasing their immunosuppressive capabilities impairing CAR T-cell efficacy. We analyzed 18F-FDG PET/CT imaging data from 63 relapsed/refractory diffuse large B-cell lymphoma receiving CAR T-cells, quantifying radiomic features including tumor sphericity and lesion aggressiveness through standardized uptake values (SUV). Statistical analyses revealed significant correlations between these metrics and progression-free survival (PFS), emphasizing that individual lesions with complex morphology and elevated metabolism play a critical role in shaping long-term treatment outcomes. We demonstrated the potential of using data-driven mathematical models in finding molecular-imaging based biomarkers to identify lymphoma patients treated with CAR T-cell therapy having higher risk of disease progression.